In modern precision agriculture, fertigation has become a cornerstone for delivering nutrients efficiently and accurately to crops. Yet, behind every successful fertigation program lies a critical – and sometimes underestimated – step: the preparation of fertilizer solutions. Errors at this stage can lead to precipitation, emitter clogging, and nutrient imbalances, ultimately reducing Nutrient Use Efficiency and crop performance.
This guide explores how to manage fertilizer compatibility, optimize solubility, and prepare stable stock solutions that support both crop productivity and irrigation system integrity.
Water Quality: The Starting Point
Every successful fertigation program begins with an understanding of water quality. Parameters such as bicarbonate concentration, electrical conductivity, and baseline pH have a direct impact on fertilizer behavior in solution. These factors influence not only solubility and stability but also the need for acidification and the overall fertigation strategy. Taking the time to analyze irrigation water provides a solid foundation for accurate and reliable solution preparation.
Why Separate Fertilizers into Different Tanks?
The separation of fertilizers into multiple tanks is not merely a technical preference – it is a chemical necessity.
Certain nutrient combinations are incompatible when dissolved together at high concentrations. The most common and problematic reaction occurs between calcium ions and phosphates or sulfates. When these elements are combined in a concentrated solution, they tend to form insoluble compounds such as calcium phosphate or gypsum. These precipitates settle out of the solution and may later cause clogging in irrigation systems.
To avoid such reactions, fertilizers are typically divided between two stock tanks. Calcium-based fertilizers, such as calcium nitrate, are placed in one tank together with nitrates, while phosphates, potassium fertilizers, magnesium, and sulfates are prepared in a separate tank. This simple but essential separation ensures chemical stability at the stock solution level.
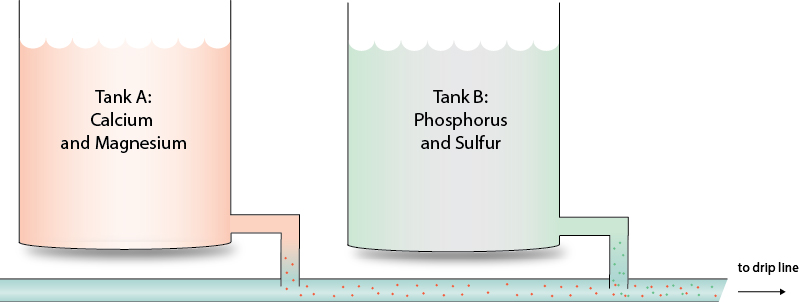
What Happens in the Drip Lines?
Although fertilizers are kept separate in stock tanks, they are eventually injected together into the irrigation system. At this stage, however, the situation is very different. The solutions are highly diluted, and the concentration of reactive ions is significantly lower. Under these conditions, the risk of precipitation is minimal, provided that the system is properly managed. This explains why separation is critical in the tanks, yet safe mixing occurs downstream during fertigation.
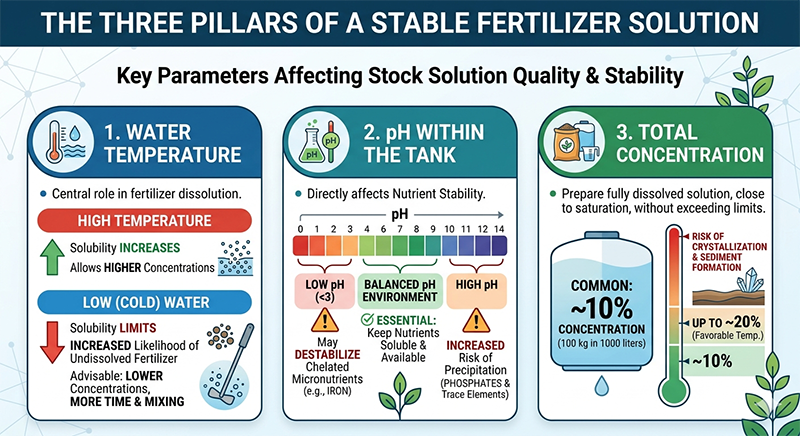
The Three Pillars of a Stable Fertilizer Solution
The quality and stability of a stock solution depend on three key parameters: temperature, pH, and total concentration.
Water temperature plays a central role in determining how much fertilizer can dissolve. As temperature rises, solubility increases, allowing higher concentrations to be achieved. In contrast, cold water limits solubility, increasing the likelihood of undissolved fertilizer that practically doesn't reach plants. In such conditions, it is advisable to work at lower concentrations and allow more time and mixing to ensure full dissolution.
The pH within the tank is equally important, as it directly affects nutrient stability. Low pH levels, particularly below 3, may destabilize chelated micronutrients such as iron, while higher pH levels increase the risk of precipitation of phosphates and certain trace elements. Maintaining a balanced pH environment is therefore essential to keep nutrients soluble and available to plants.
Total concentration is the third critical factor. In general, the objective is to prepare a solution that is fully dissolved and close to saturation, without exceeding solubility limits. In practice, a concentration of around 10% (i.e., 100 kg fertilizer in 1000 liter of water) is commonly used. Higher concentrations – up to approximately 20% – are possible under favorable temperature conditions. Exceeding these limits increases the risk of crystallization and sediment formation at the bottom of the tank.
Proper Addition of Micronutrients
Micronutrients, especially when supplied in chelated form, require careful placement within the tank system. Iron chelates, for example, are generally best added to the calcium tank. This recommendation is linked to pH considerations. The tank containing phosphates is often acidified, sometimes reaching very low pH levels that can destabilize chelates and render the micronutrients unavailable to plants. By placing iron chelates in the calcium tank, where pH conditions are typically more moderate, their stability and effectiveness are better preserved.
💡Tip: micronutrient dissolution can be managed easily and safely by using Poly-Feed NPK formulas. These fertilizers, containing chelated micronutrients, should be dissolved separately from the calcium and magnesium fertilizers.
Practical Aspects: Preparing a Stable Solution
Preparing fertilizer solutions is a precise operation that requires attention to detail and proper sequencing.
Correct Preparation Procedure:
- Fill the tank with 50–70% of the required water volume
- Start continuous mixing (mechanical agitator or recirculation pump)
- Gradually add fertilizers – starting with the least soluble products
- Once fully dissolved, complete filling to the final volume
It is important to emphasize that fertilizers should never be added without active mixing, as this can lead to localized over-concentration and incomplete dissolution.
Explore various tools for fertilizer dissolving
Clean, covered tanks are recommended to prevent contamination and algae development, while protection from direct sunlight helps preserve the stability of sensitive components such as chelates.
Summary: Precision Starts in the Tank
The preparation of fertilizer solutions is the foundation of effective fertigation. Mistakes at this stage can compromise the entire system, affecting both nutrient availability and irrigation performance.
A properly prepared solution supports optimal nutrient availability, improves Nutrient Use Efficiency, and ensures the reliable operation of irrigation systems without clogging.
Using high-quality fertilizers that are clean, fully soluble, and consistent in composition is essential for achieving these goals. Advanced tools such as NutriNet™ can further support growers by designing precise fertigation programs that take into account compatibility, dissolution, and optimal tank allocation.
Ultimately, mastering tank mixes is not just about avoiding technical problems – it is about enabling crops to reach their full potential while maintaining the efficiency and longevity of the irrigation system.
Download the full for guide for preparation of fertilizer solutions




